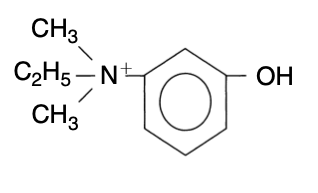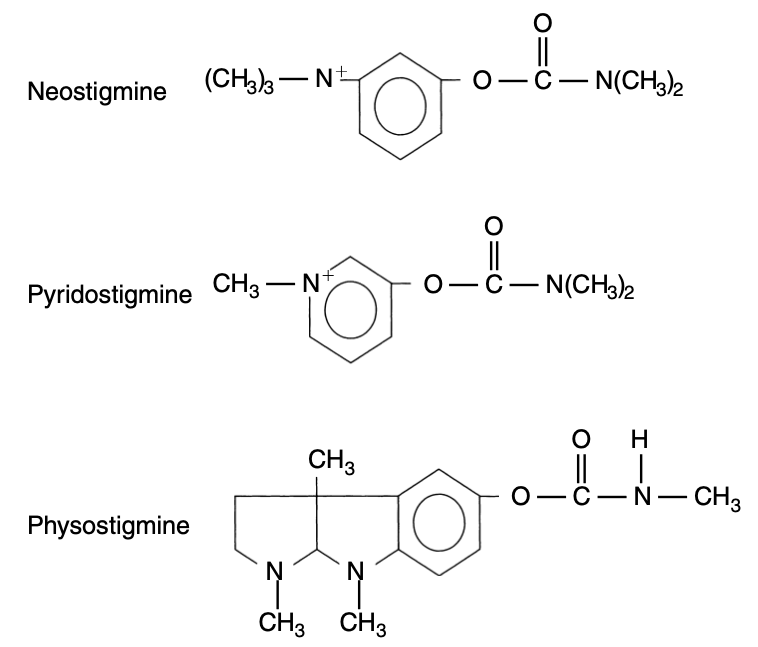
Adapted from E-learning for Health
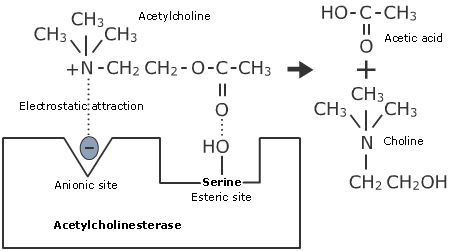
- Rapidly hydrolyses acetylcholine into choline and acetic acid
- Contains two binding sites:
- The Anionic site
- The Esteratic site
- The Anionic binding site contains a negatively charged glutamate
- This binds reversibly via electrostatic attraction to the positively charged quaternary amide on the choline portion of acetylcholine
- The Esteratic binding site contains serine amino acids that hydrolyse acetylcholine
- The enzyme is temporarily acetylated
- Choline is reabsorbed via presynaptic transporters, where it is recycled