- Primary and secondary prevention in stroke/myocardial infarction
- Reduces the risk of unstable angina progressing to MI
- Reduces mortality following MI
- Reduces risk of stroke in those who've had a TIA
- Analgesic/anti-inflammatory
Aspiring
Aspiring
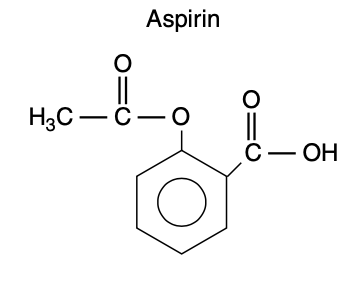
- Aspirin (acetylsalicylic acid) is an aromatic ester of acetic acid
- At low dose (75mg/day):
- Irreversibly, selectively inhibits platelet COX via enzyme acetylation
- Preserves vessel wall COX
- This reduces production of TXA2 and therefore reduces thromboxane-induced platelet aggregation and vasoconstriction
- Vessel-wall prostacyclin synthesis is unaltered and therefore vasodilation occurs
Absorption
- Weak acid with a pKa of 3
- Therefore essentially wholly unionised in the stomach, facilitating absorption
- However more drug is overall absorbed in the ileum/small bowel due to its increased surface area
- Acetylsalicylate and salicylate ions may be trapped in the alkaline environment of the mucosal cells
- Unable to reach systemic circulation, therefore increased propensity to cause local effects
- 65-70% oral bioavailability
Distribution
- 85% protein bound (mostly to albumin as a weak acid)
Metabolism
- Rapidly hydrolysed by intestinal and hepatic esterases to salicylate and acetic acid
- Further hepatic metabolism:
- Glycine conjugation to salicyluric acid
- Glucuronide derivatives
- Elimination half life is short (15-30mins) if obeying 1st order kinetics
- Significantly prolonged if glycine conjugation becomes saturated (as in overdose) → zero order kinetics
Excretion
- Salicylate and metabolites are excreted in the urine
- Excretion is enhanced under alkaline conditions
- Generic NSAID effects
Metabolic effects
- Effect on the metabolic state are of little significance, except in aspirin overdose i.e. salicylate poisoning
- Uncouples oxidative phosphorylation
- This increases oxygen consumption and carbon dioxide production
- Initially minute ventilation increases to keep PaCO2 static
- When aspirin levels increase there is direct stimulation of the respiratory centre to cause respiratory alkalosis
- Picture complicated by concurrent metabolic acidosis
- (NB in children, rising aspirin levels depress the respiratory centre, causing mixed respiratory/metabolic acidosis)
Reye's syndrome
- An uncommon syndrome that mainly occurs in children
- Leads to:
- Widespread mitochondrial damage
- Fatty change in the liver and hepatic failure
- Encephalopathy and cerebral oedema
- Overall mortality 40%
- Aspirin should be avoided in those <16yrs old (except in specific indications e.g. JIA)