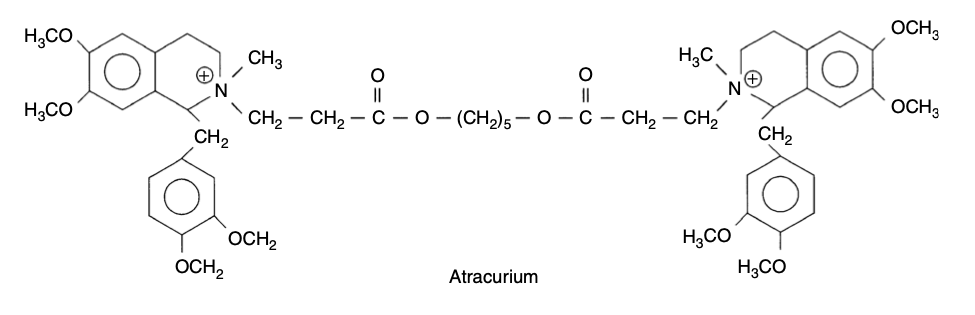
- Atracurium is a benzylisoquinolinium NMBA formed as a mixture of ten stereoisomers, as it has four chiral centres
- Colourless solution of 10mg/ml stored at 4'C (i.e. fridge) and pH 4.0
- It is formed as atracurium besylate and is therefore a base
- Bases are formed as drug-sulphate/similar e.g. morphine sulphate, bupivacaine hydrochloride, atracurium besylate
- Acidic drugs are formed as sodium-drug e.g. sodium diclofenac, sodium thiopental
- Single dose 0.5mg/kg provides intubating conditions in 90-120s
- Can be used as an infusion to maintain neuromuscular blockade on intensive care
- Is metabolised by two pathways:
- Ester-hydrolysis (60%)
- Hoffman elimination (minor pathway, 40%)
- Non-specific esterases hydrolyse atracurium into:
- Monoquaternary alcohol
- Quaternary acid
- Laudanosine
- Is accelerated by acidic conditions although changes within the clinical range probably don't affect rate of ester hydrolysis
- Spontaneous breakdown in a temperature- and pH-dependent fashion into:
- Monoquaternary acrylate (can cause seizures)
- Laudanosine
- Process is slowed by acidosis and hypothermia
- Is a glycine antagonist
- Is a tertiary amine
- Can cause seizures (in concentrations in excess of those encountered clinically)
- No neuromuscular blocking properties
- Rapidly eliminated by renal excretion
- Associated with histamine release, which may cause bronchospasm, hypotension and other anaphylactoid symptoms
- Critical illness myopathy
- Cis-atracurium is one of the ten stereoisomers in atracurium
- Safe for use from 2yrs upwards
- Presents as a colourless solution in 2mg/ml or 5mg/ml concentrations and is stored at 4'C
- Used for induction and maintenance of muscle paralysis at a dose of 0.15 - 0.2mg/kg
- Is 3-4x more potent than atracurium and therefore has a slower onset time
- The onset time can be reduced by increasing the dose
- Cis-atracurium has a low potential for histamine release which makes this viable
- Does not undergo ester hydrolysis
- Metabolism is 100% by Hoffman degradation
- All metabolites are void of neuromuscular blocking properties
- No changes to kinetic profile in end-stage renal or hepatic failure
