- Barbiturates are derived from barbituric acid, a condensation of malonic acid and urea

- Barbituric acid has a pyrimidine ring nucleus
- It is not a CNS depressant
- As an oxygen molecule is bonded at the C2 carbon, barbituric acid is an oxybarbiturate
- Oxybarbiturates are poorly lipid soluble and poorly protein bound
- They are excreted almost entirely unchanged in the urine
- When a sulphur group is exchanged in for an oxygen at C2, oxybarbiturates become thiobarbiturates
- Thiobarbiturates are:
- Highly lipid soluble and protein bound
- Completely metabolised in the liver
- This provides them with a fast onset of action, yet also a short duration of action and short recovery period
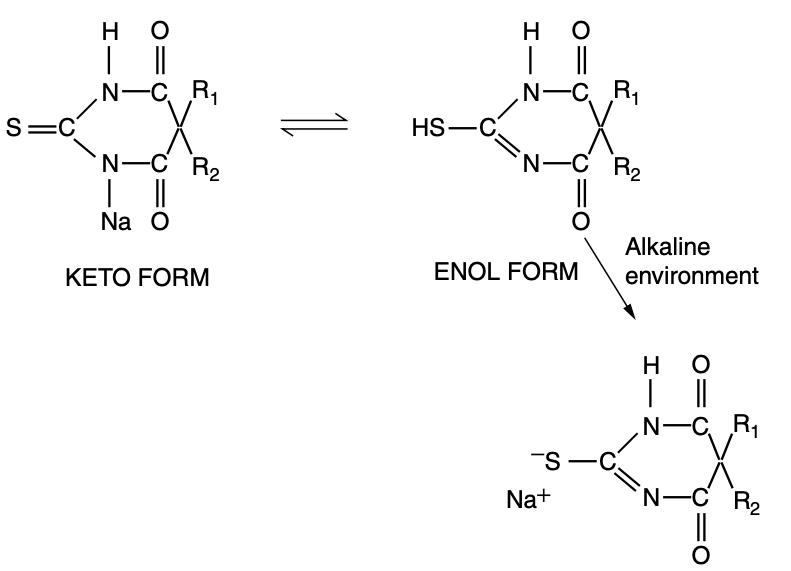
- Thiobarbiturates are poorly water soluble at neutral pH, existing in their keto form
- In order to become water soluble, it is necessary for them to exist in their enol form
- This process is favoured by alkaline environments
- This is an example of dynamic structural isomerism - tautomerisation
- Barbiturates facilitate inhibitory synaptic transmission by:
- Agonising the GABAA receptor by binding to the GA site on the β subunit
- Antagonising AMPA and kainite (non-NMDA) glutamate receptors, inhibiting excitatory post-synaptic transmission
- Antagonising neuronal nAChR and K+ channels
- They also provide a neuroprotective effect by:
- Antagonising voltage gated sodium and calcium channels
- Scavenging free radicals

