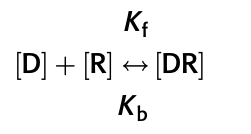
D + R ⇄ DR
- When a drug (D) binds a receptor (R) it forms a drug-receptor complex (DR)
- It is assumed that the magnitude of the response is proportional to the concentration of DR
- The law of mass actions states that the rate of the reaction is proportional to the concentration of the reacting components
- Where:
- Kf is the rate of the forward reaction
- Kb is the rate of the backward reaction
- [D], [R] and [DR] are the concentrations of the drug, receptor and drug-receptor complex respectively
- It is a specific, saturable and concentration-dependent interaction