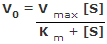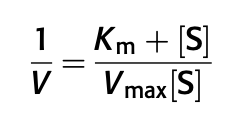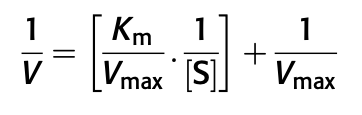- An enzyme is a catalyst that increases the velocity of a chemical reaction without itself being consumed in the reaction
- The rate of a reaction is therefore dependent on the concentration of substrate [S] present and the presence of an enzyme [E]
E + S ⇄ ES ⇄ P
- I.e. an increase in substrate concentration will increase reaction velocity
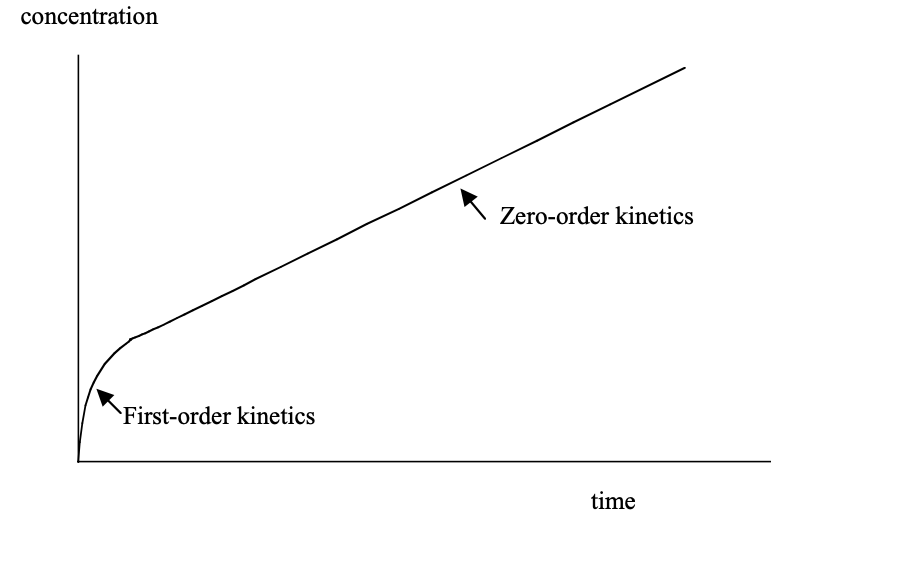
- This can be plotted as a graph of reaction velocity (V0) vs. substrate concentration (S)
- The graph produced is an inverted rectangular hyperbola, and is appropriate to describe most enzyme reactions
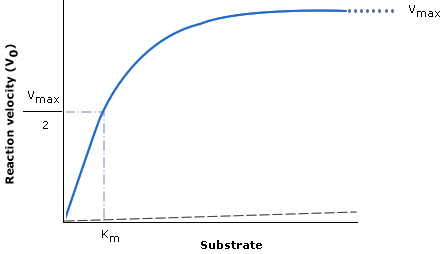
- KM is the substrate concentration at which reaction velocity is half of maximal value
- It changes according to the affinity of the enzyme for the substrate
- Higher affinity = lower KM
- It is equivalent for ED50 in drug dose-response curves
- The portion of the curve to the left of KM exhibits first-order kinetics
- The far right portion of the curve (horizontal) exhibits zero-order kinetics
The Michaelis-Menton Equation
- The Michaelis-Menten equation describes the rate of a biological reaction according to enzyme characteristics and substrate concentration, until it reaches enzyme saturation
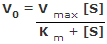
- Enzyme kinetics are first order at low substrate concentrations, V ⍺ [S]
- If [S] is low, the +[S] part of the equation trends to nothing
- Therefore V is proportional to [S] by a factor of Vmax/KM , i.e. it is the [S] that will have the effect on V
- Thus the rate depends on the concentration of the reacting components
- It is an exponential process
- Enzyme kinetics are zero order at high substrate concentrations (i.e. when the enzyme is saturated), V ⍺ Vmax
- If [S] is high, the Vmax.[S] part of the equation becomes very large
- Therefore V is proportional to Vmax i.e. V is independent of [S]
- I.e. reaction velocity is constant and independent of substrate concentration