- Is unstable when exposed to light
- Corrodes certain metals
- Dissolves into rubber and may leak into breathing circuits even after the vaporiser is turned off
- Is therefore stored with 0.01% thymol to prevent decomposition by light and liberation of free bromine
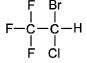
Halothane
Halothane
- Halothane is a halogenated hydrocarbon
- It has a chiral carbon
- As a volatile anaesthetic it has largely been superseded
| MAC | Blood:gas coefficient | Oil:gas coefficient | Molecular weight | Boiling point | SVP at 20ºC | Safe ppm |
| 0.8% | 2.2 | 224 | 197 | 50.2ºC | 32.3kPa | 10 |
- Non-irritant
- Sweet odour
Metabolism
- 20 - 25% oxidative metabolism by CYP2E1 enzyme, producing:
- Trifluoroacetic acid
- Bromide and chloride ions
- There is also reductive metabolism to fluoride and other reductive metabolites when there is liver hypoxia
- While these are toxic they are not thought to be involved in halothane hepatitis
Respiratory
- Some reduction in VT but overall does not affect PaCO2
- Significant bronchodilator properties and therefore useful in asthma
Cardiovascular
- Net reduction in blood pressure owing to:
- Direct myocardial depressant effect therefore significantly reduced contractility
- Indirect effects increasing vagal tone, reducing heart rate
- Reduces SVR
- Reduces splanchnic blood flow
- Significant sensitisation to endogenous and exogenous catecholamines
- Doses of adrenaline should be limited to 10ml of 1:100,000 in 10mins to reduce risk of VT
Neurological
- Massive increase in CBF causing raised ICP, but reduces CMRO2
- Burst suppression on EEG
- Some potentiation of muscle relaxation and some uterine relaxation
- No analgesic properties
Reversible hepatic damage
- Occurs in 25% of patients
- Often subclinical
- Associated with rise in transaminase levels
- Can affect metabolism of drugs used in peri-operative period
- Probably secondary to hepatic hypoxia as halothane reduces splanchnic blood flow
Halothane hepatitis
- A severe centrilobular hepatic necrosis and consequent fulminant liver failure
- Occurs in 1 in 80,000 - 200,000 children, and 1 in 2,500 - 35,000 adults
- Oxidative metabolism of halothane generates trifluoroacetyl chloride
- Trifluoroacetyl chloride acts as a hapten
- Binds covalently to hepatic proteins
- Induces antibody formation in a T-cell mediated process
- Risk factors include:
- Middle aged
- Female
- Liver disease
- Hypoxia
- Obesity
- Repeated exposure (avoid halothane if administered within previous 3 months)
- Previous reaction
- Mortality 50 - 75%