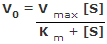Direct, reversible inhibition (competitive antagonism)
- The degree of enzyme inhibition is dependent on the concentration of the competitive antagonist vs. the concentration of the natural agonist
- There are a multitude of examples:
- Neostigmine - AChE inhibitor
- Milrinone - PDE inhibitor
- Ramipril - ACE inhibitor
- NSAIDs - COX inhibitor
Direct, irreversible inhibition
- Drugs have a long duration of action as they require re-synthesis of the enzyme before normal activity can be resumed
- Examples include:
- Aspirin acetylation of COX
- Phenelzine and tranylcypromine, which are non-selective MAO inhibitors
- Action of intermediary messengers to reduce enzyme activity
- Drugs that demonstrate indirect enzyme inhibition may be:
- Agonists at G-protein coupled receptors linked to Gi alpha subunits, which reduces intracellular cAMP e.g. clonidine, opioids (MOP)
- Antagonists at G-protein receptors linked to Gs alpha subunits, which inhibits increases in intracellular cAMP e.g. β-blockers
Adapted from Physics, Pharmacology and Physiology for Anaesthetists
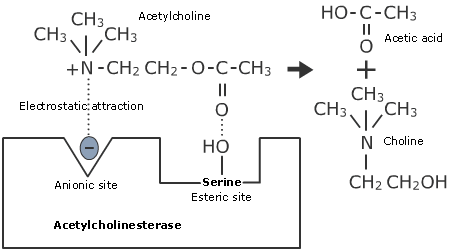
Reversible, competitive anionic site inhibitors
- E.g. edrophonium
- Short-acting inhibitors that are used for diagnostic purposes
Reversible enzyme carbamylators
- E.g. neostigmine, physostigmine, pyridostigmine
- Bind to both esteratic and anionic sites
- Carbamylate the enzyme, which reacts with water more slowly
- This reduces the rate of ACh breakdown, increasing concentrations in the synaptic cleft
- As such they increase ACh concentration to reverse the effect of the competitive, non-depolarising nAChR antagonists
- Patients with myasthenia gravis take pyridostigmine
- E.g. organophosphates, certain biochemical agents
- Interact with the esteratic site to phosphorylate the enzyme
- The enzyme becomes 'aged' with time
- The phosphorylated enzyme reacts with water even more slowly, causing ACh concentrations to rise centrally and peripherally, leading to cholinergic crisis
- Pralidoxime (if used before 36-48hrs post-exposure) displaces the phosphate from the esteratic site and remains bound, allowing clearance of the poison
- Without treatment, recovery is dependent on new AChE synthesis