| MAC | Blood:gas coefficient | Oil:gas coefficient | Molecular weight | Boiling point | SVP at 20ºC | Safe ppm |
| 1.2% | 1.4 | 98 | 184.5 | 48.5ºC | 33.2kPa | 50 |
- Irritant
- Pungent smell
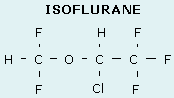
Isoflurane
| MAC | Blood:gas coefficient | Oil:gas coefficient | Molecular weight | Boiling point | SVP at 20ºC | Safe ppm |
| 1.2% | 1.4 | 98 | 184.5 | 48.5ºC | 33.2kPa | 50 |
Metabolism
Respiratory
Cardiovascular
Neurological