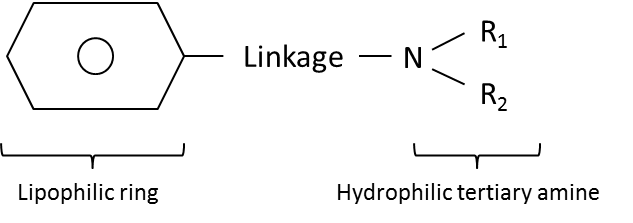
- Local anaesthetics are all weak bases
- All are formed from a lipophilic ring (aromatic group) and a hydrophilic tertiary amine that are linked:
- Increasing the length of the carbon chain at any point in the molecule increases its lipid solubility, potency and duration of action
- The linkage can be either:
- An ester linkage
- Examples include amethocaine (tetracaine), procaine and cocaine
- Esters are comparatively unstable in solution
- An amide linkage
- Examples include lidocaine, prilocaine, chloroprocaine, (levo)bupivacaine and ropivacaine
- Amides have a long shelf-life of approximately 2yrs