The phenomenon where the rise in alveolar partial pressure of nitrous oxide is disproportionately rapid when administered in high concentrations
- The concentration effect arises because the solubility of nitrous oxide in blood is 20x greater than that of nitrogen
- N2O in the alveolus is rapidly absorbed into the bloodstream, whereas nitrogen is slow to diffuse out of the bloodstream
- Therefore:
- The alveolus decreases in volume, increasing the partial pressure of N2O
- More gas from the airways is drawn into the alveolus to replenish the lost volume
- If this gas contains volatile anaesthetic, it will increase the concentration of the volatile agent in the alveolus
- This can be demonstrated by the wash-in curve, a graph of FA/FI ratio vs. time:
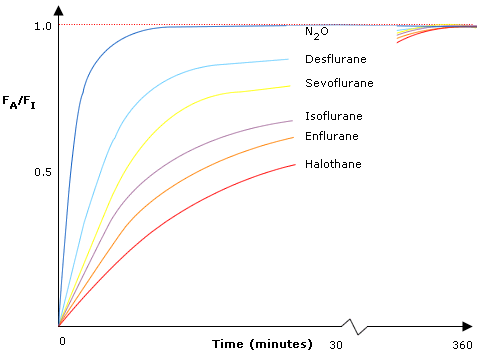
- It is a negative exponential curve, as the rate of change decreases with time
- The more insoluble the agent (blood:gas partition coefficient), the faster the rate of onset and the steeper the curve
- However, even though N2O (B:G 0.47) has a greater blood:gas partition coeffecient than desflurane (B:G 0.4) it has a more rapid onset - because of the concentration effect
- Eventually all the lines will plateau at an FA/FI ratio of 1.0
- Limitations of this model:
- Assumes nitrous oxide is the only gas absorbed
- Assumes that 50% is absorbed from alveolus to pulmonary capillaries
- Doesn't include the effects of nitrogen leaving the body
- Assumes that the volume deficit from rapid nitrous absorption is replaced by gas of the same concentration as that initially inspired
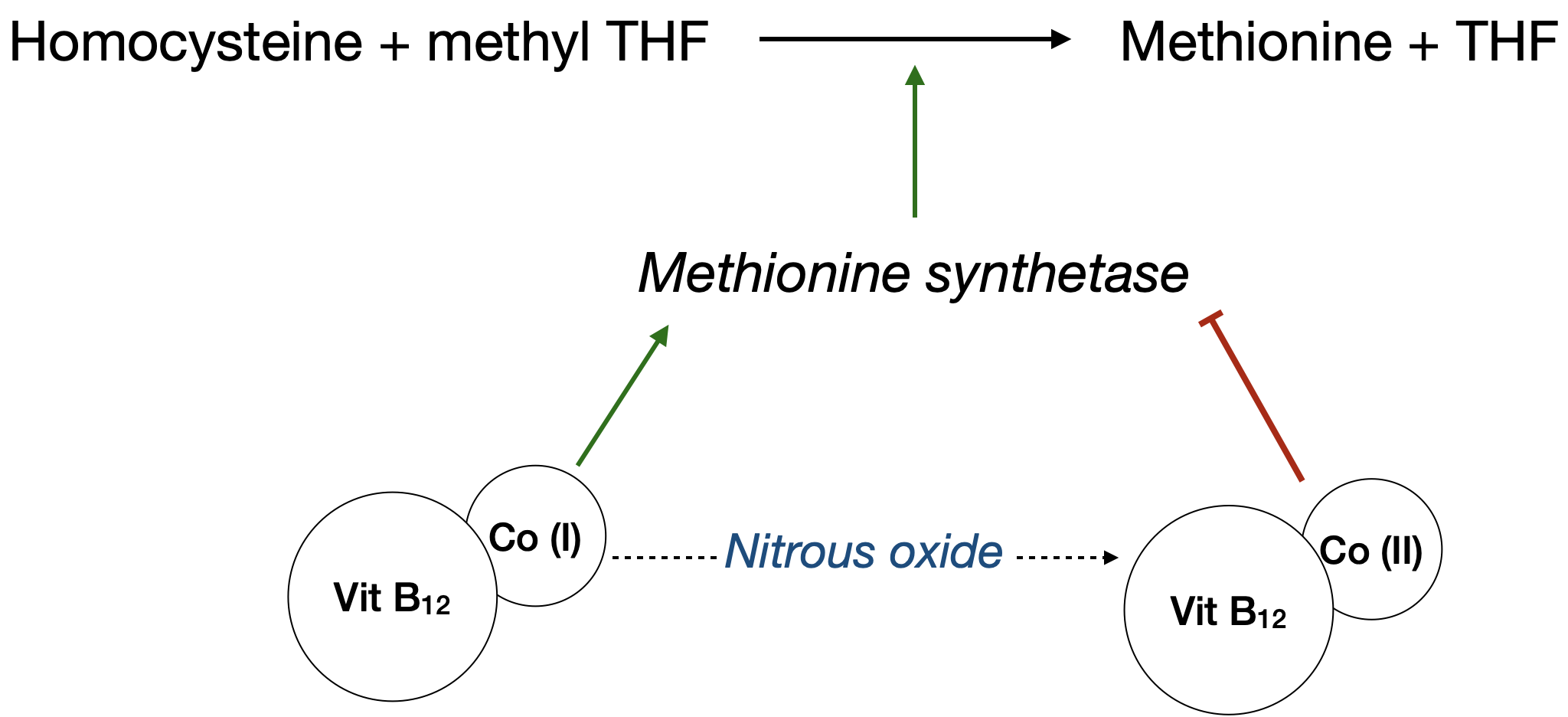
The phenomenon whereby the speed of onset of an inhalational anaesthetic agent is increased when it is used with nitrous oxide as a carrier gas
- This is a consequence of the concentration effect
- Increases alveolar concentration of oxygen and volatile agents quickly
- Reduces induction time
- The reverse of the concentration effect
- At the end of anaesthesia, nitrous oxide/oxygen in the blood stream is replaced by air (nitrogen/oxygen)
- The volume of nitrous oxide diffusing from pulmonary capillaries to alveolus is high
- It is greater than the volume of nitrogen diffusing from alveolus to pulmonary capillaries
- As such it dilutes alveolar oxygen concentration and may cause hypoxia
- Prevented by changing inhaled gas mix to 100% oxygen