- Opioids have multiple negative impacts on respiratory function which can potentially lead to severe respiratory depression:
- Depression of the respiratory centre in the brainstem, disrupting the generation of the normal respiratory rhythm and reducing alveolar minute ventilation
- Reduced oropharyngeal muscle tone, resulting in upper airway obstruction
- Negatively affect both central and peripheral chemoreflex loops
- Depression of the hypothalamus, leading to sedation via increased arousal thresholds
- These effects are primarily related to MOP receptor agonism within the respiratory network (pons, medulla) and carotid bodies (peripheral chemoreceptors)
Progress of respiratory changes following opioid overdose
- Follows a stereotypical pattern
- Irregular breathing
- Periodic or cyclic breathing
- Gasping
- Full cessation (i.e. apnoea)
- In some cases abrupt apnoea occurs without other warning signs
- Apnoea leads to asphyxia (low arterial oxygen tension but high arterial carbon dioxide tension) and potentially cardiac arrest
- Opioids affect multiple areas influencing control of ventilation:
- The pre-Botzinger complex (medulla) which is essential for respiratory rhythm generation
- Parabrachial complex (the Killiker-Fuse and parabrachial nuclei), which sustains upper airway patency, integrates sensory input from chemoreceptors and adjusts ventilation in response to arterial oxygen and carbon dioxide levels
- Phrenic pre-motor and motor neurons; a direct effect at high opioid doses which depresses phrenic motor output
Effects of opioids on ventilatory control; the hypercapnic ventilatory response (HCVR)
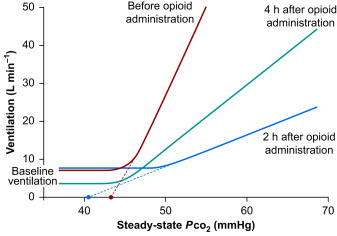
Adapted from Opioid-induced respiratory depression (BJA Education, 2024)
- The HCVR (i.e. response to inhaled carbon dioxide) is a sensitive measure of the effects of opioids
- In the absence of opioids, a the HCVR graph (of steady-state PCO2 vs. ventilation) is 'hockey-stick' shaped
- The flat part of the curve demonstrates CO2-insensitive ventilation, i.e. baseline ventilation before a 'ventilatory recruitment threshold' is reached
- The linear, increasing part of the curve is the HCVR; it is described by both a slope (i.e. gradient) and an apnoeic threshold
- Opioids effect the HCVR graph by:
- A (slow) decrease in baseline ventilation
- Shifting the curve to the right i.e. higher levels of PCO2 are required to trigger the ventilatory recruitment threshold
- A reduction in the gradient of the HCVR i.e. less potent ventilatory response in the presence of opioids
