- Presents as the pure S-enantiomer in 0.2%, 0.75% and 1% solutions
- The R-enantiomer is less potent and more toxic
- Not prepared with vasoconstrictors as these don't alter its duration of action or uptake from the tissues
Ropivacaine
Ropivacaine
- Ropivacaine is an amide local anaesthetic which contains a chiral carbon
- Has a butyl group on its piperidine nitrogen
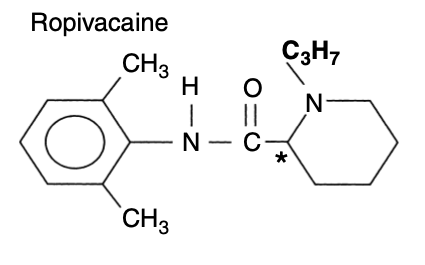
- Compared to bupivacaine, ropivacaine has:
- An enantiopure preparation
- Reduced lipid potency
- An improved toxicity profile (Acta Biomed, 2008)
- The reduced lipid solubility means there is less penetration of large-diameter, myelinated Aβ motor neurones
- This causes relative sparing initially (though will become blocked during continuous infusion)
- Motor block is therefore slower in onset, less dense and shorter in duration
Absorption
- pKa 8.1
- Means only 15% unionised at physiological pH, giving it a slow onset time
Distribution
- 94% protein bound
- Relative lipid solubility vs. cocaine: 300x
- Toxic doses:
- Plasma concentration: >4μg/ml
- Dose: 2mg/kg (unchanged by adrenaline)
Metabolism
- Hepatic aromatic hydroxylation to the weakly active metabolites 3-hydroxy- and 4-hydroxy-ropivacaine
Excretion
- Elimination half-life 120mins