| MAC | Blood:gas coefficient | Oil:gas coefficient | Molecular weight | Boiling point | SVP at 20ºC | Safe ppm |
| 1.8% | 0.7 | 80 | 200.1 | 58.5ºC | 22.7kPa | No set limit |
Sevoflurane
Sevoflurane
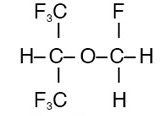
- Sevoflurane is a polyfluorinated isopropyl methyl ester
- It is an achiral molecule
- SEVoflurane has SEVen fluoride ions
One pot method
- All the ingredients to make sevoflurane are mixed, then water added to 300ppm
Chloro-fluoro method
- The basic molecular architecture is created but with chlorine molecules
- Fluorine is then substituted for chlorine to produce sevoflurane
- A Lewis acid is any substance that can accept an electron pair
- It includes many metal oxides, H+ and glass
- Sevoflurane is formulated with 300ppm water as this is an inhibitor of Lewis acids
- If the concentration of water is <100ppm, the Lewis acids attack at sevoflurane's ether and halogen bonds
- This releases the highly toxic substance hydrofluoric acid
- Hydrofluoric acid can also corrode glass, exposing the sevoflurane to further Lewis acids
- Sevoflurane is therefore kept in a polyethylene naphthalate bottle
- A dry formulation containing <130ppm water is available, and kept in an aluminium bottle with an epoxyphenolic resin lacquer
Metabolism
- 3.5 - 5% metabolised by CYP2E1 subfamily to:
- Organic fluoride - hexafluroisopropanol (HFIP)
- Inorganic fluoride ions (renal toxicity)
- It is not metabolised to trifluoroacetic acid therefore not linked with hepatitis
Respiratory
- Non-irritant with a pleasant odour
- Dose dependent respiratory depression
- Reduces VT to a greater extent than it stimulates respiratory rate, and therefore there is a rise PaCO2
- Relaxes bronchial smooth muscle via histamine/acetylcholine
Cardiovascular
- Reduces vascular resistance: SVR, coronary and cerebral circulations
- Blood pressure is therefore reduced
- There is, however, no change to splanchnic blood flow
- No catecholamine sensitisation
- Can lengthen QTc
Neurological
- Increases CBF and reduces CMRO2
- Potentiates muscle relaxation, including some relaxation of the uterus
- Can cause burst suppression on EEG
- Purportedly some analgesic properties
- Incidence of excitatory phenomenon on gas induction/waking most common with sevoflurane
- In the presence of CO2 absorbers, sevoflurane forms compounds A-E
- Only compound A and B are present in sufficient amount to analyse
- Formation is favoured in KOH-based absorbents (vs. NaOH), especially when dry
- Reaction releases heat and consumes sevoflurane
- Compound A
- A tri-fluoro-methyl vinyl ester
- LC50 of compound A in rats is 300-400ppm after 3hrs
- This suggests human nephrotoxic threshold of 150-200ppm
- However, with flow rates of 0.25L/min for 5hrs, peak compound A concentration <20ppm and no change to renal function
- Humans also have 20x reduced levels of β-lyase vs. rats and therefore there is reduced conversion of compound A to toxic metabolites
- Evidence demonstrates sevoflurane use does not increase risk of renal injury (BJA, 2022)
- However some concern it may cause nephrogenic diabetes insipidus if used for prolonged sedation on ICU (BJA, 2022)