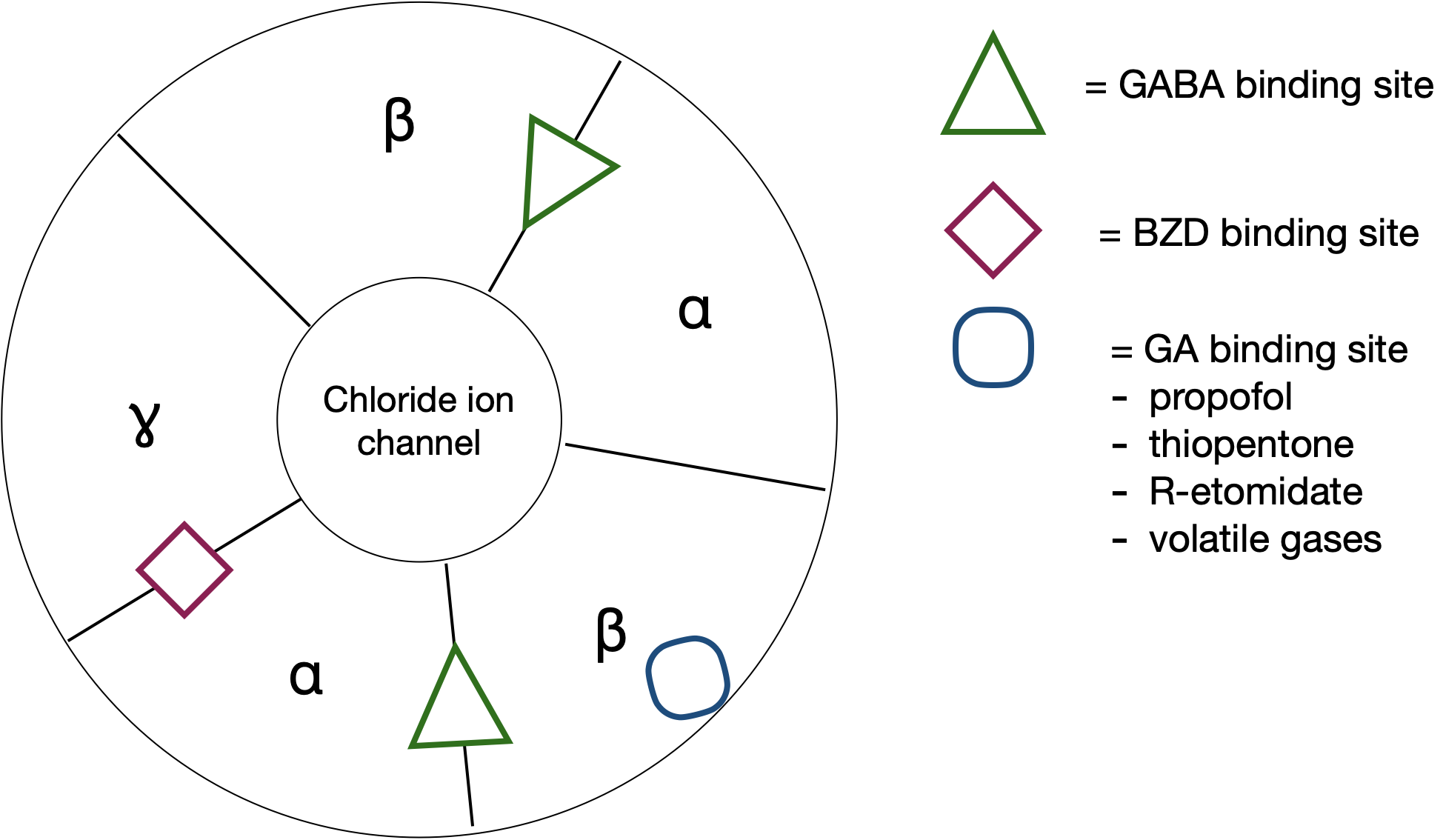
- The Meyer-Overton Hypothesis proposed that the potency of an anaesthetic agent is related to its lipid solubility
- Potency itself is described by minimum alveolar concentration (MAC)
- Lipid solubility is described by the oil:gas partition solubility coefficient
- The hypothesis states that once a certain number of anaesthetic molecules were dissolved in the lipid bilayer of CNS cells, it would disrupt membrane and thus cellular function
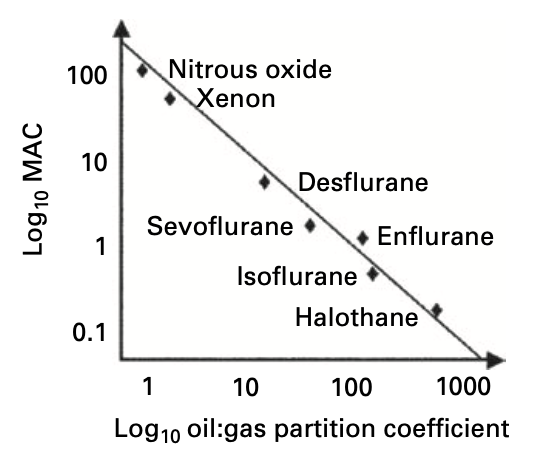
- The correlation between lipid solubility and potency suggested a non-specific mechanism of action based on physicochemical properties
- Problems with the theory include:
- Ketamine being an extreme outlier
- R-etomidate has anaesthetic activity though its enantiomer S-etomidate (identical lipid solubility) does not
- Later interpretation pointed to any highly lipophilic area being a potential site of action (e.g. cell membrane)
MAC
- The minimum alveolar concentration of an anaesthetic vapour at equilibrium to prevent movement to a standardised surgical stimulus in 50% of un-premedicated patients at sea level (1atm)