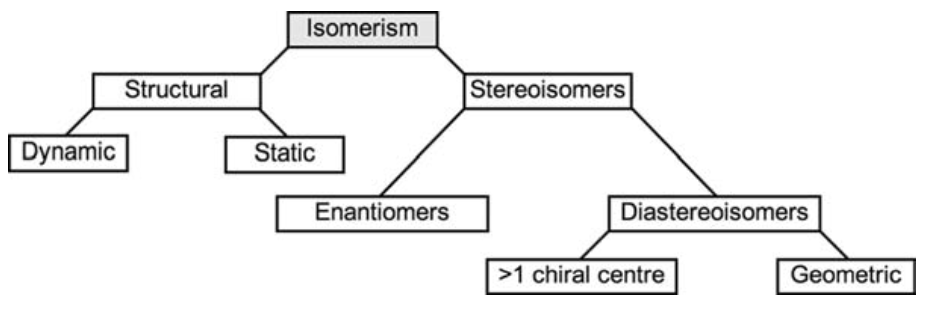
- Stereo-isomerism describes molecules with identical chemical formulae and order of atomic bonds, but with a different 3D configuration
Geometric (cis-trans) stereo-isomerism
- Cis-trans isomers are compounds with two dissimilar groups attached either side of a double-bond (or ring) e.g. alkenes, which form isomers because of the immobility of the double-bond/ring
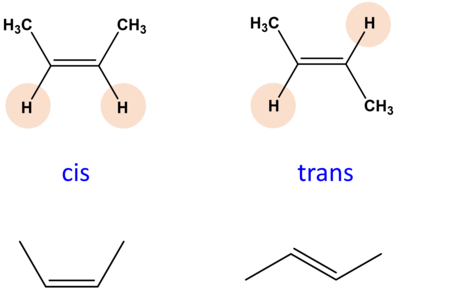
- The groups can be arranged either diagonally across the bond (trans) or on the same side of the bond (cis), each leading to different chemical properties
- The classic example is the neuromuscular blocking agent mivacurium, which presents as a mixture of its stereoisomers:
- 58% trans-trans isomer
- 36% cis-trans isomer
- 6% cis-cis isomer, which has 10% potency but 10x the half-life of the other stereoisomers
Optical stereo-isomerism (enantiomers)
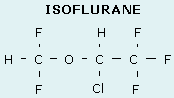
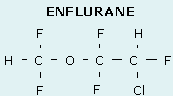
- Optical stereo-isomers arise owing to the presence of a chiral centre, around which other atoms may be placed into two non-superimposable configurations
- A chiral centre is an atom bound to four dissimilar groups (typically a carbon or quaternary nitrogen); it is sometimes also called an asymmetrical carbon
- Molecules which contain a chiral centre are called enantiomers
Classification of enantiomers
- Enantiomers were historically differentiated based on their ability to rotate polarised light in different directions
- Molecules were either dextro-rotatory (D) or levo-rotatory (L)
- D-enantiomers rotate polarised light to the right
- L-enantiomers rotate polarised light to the left
- It remains the nomenclature for the orientation of the atomic structure of sugar and amino acid molecules (e.g. dextrose is D-glucose, levobupivacaine is L-bupivacaine)
- A more modern classification is in to rectus (R) or sinister (S) molecules
- The smallest molecular weight group around a chiral centre is arranged as being behind the plane of the page
- The remaining three groups are arranged in the plane of the page
- If the remaining groups' molecular weights decrease in a clockwise fashion it is the R(-) isomer
- If the remaining groups' molecular weights decrease in an anti-clockwise fashion it is the S(+) isomer
- For example S-ketamine, which is more potent and causes less emergence delirium compared to R-ketamine
- Diastereoisomers are molecules with more than one chiral centre; if there are X chiral centres then there are X2 possible diastereoisomers
- The classic example is atracurium, which has four chiral centres and ten possible stereoisomers
Enantiopure and racemic mixtures
- A racemic mixture describes a mixture with two enantiomers present in equal proportions, and will not rotate polarised light
- A number of drugs are formulated as racemic mixtures e.g. ketamine, isoflurane, adrenaline, warfarin
- By comparison, an enantiopure solution is a solution where only enantiomer is present
- They are often more expensive to manufacture, but typically benefit from avoiding negative effects associated with a certain enantiomer
- For example:
- Enantiopure S-ketamine is more potent and associated with less emergence delirium than racemic ketamine
- Enantiopure S-bupivacaine (a.k.a levobupivacaine) is thought to be less toxic than racemic bupivacaine
- Adrenaline usually presents as L-adrenaline (15x more potent), with racemic adrenaline (racepinephrine) only being available in North America